What is a Litmus Test?
A litmus test originally meant a simple method to determine whether something was acidic or basic. It then morphed into an expression meaning a test that gives a simple yes or no answer. It turns out that a real litmus test isn’t always so “black and white”.
Litmus Paper-Red or Blue?
When we think of litmus paper, the colors red and blue come to mind. It turns out there is also a gray area, or purple, to be more accurate. The color of litmus is only solid red below pH 4.5 and solid blue above pH 8.3. From pH 4.5 to pH 8.3 the color goes through shades of purple as you can see in the top strip in the image below.

The top strip was dipped in acid pH<4.5 and the other end was dipped in base > pH8.3. The lighter purple in the middle is neutral pH 7. The bottom strip’s central section has no litmus in it all. (Image source: Wikipedia)
You can test this further with some of the liquids in the chart below. You will see that litmus paper can only give a definitive yes or no for acid/base for some of them.
pH of Things Around the House
You can do a litmus test using the liquids listed below. You will see for yourself that litmus paper test can only confirm the most acidic or basic ones but not the ones closer to neutral pH 7

Litmus Paper Tests
Drop lemon juice onto red litmus paper, nothing happens. Drop milk of magnesia onto blue litmus paper, again, nothing happens. Switch the color of the papers, repeat, and observe the change.
An interesting feature of litmus is that the color change is reversible. Drop lemon juice onto the milk of magnesia spot and milk of magnesia onto the lemon juice spot and see what happens.
Try this with different liquids. What happens if you make a spot with a weak base & then drop a strong acid on it. Or, a strong base & then a weak acid? There are all sorts of possibilities.
If you have an artistic streak, you can evoke your inner Jackson Pollock and create weird and wonderful patterns like those below on the 8×10″ (200x250mm) sheets of litmus paper.

2 large sheets of litmus, 1 red & 1 blue are compared. The one on the right is red litmus sprayed with an alkaline solution. The one on the left is red litmus sprayed with acid.
Litmus Test Chemistry
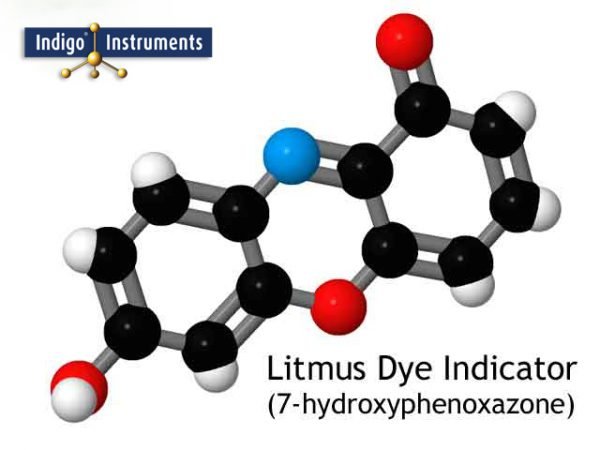
So why does litmus behave this way? It’s because of 7-hydroxyphenoxazone. When exposed to acids below pH 4.5, the molecule looks like the image below and this gives litmus paper its red color. As the pH of the test solution moves towards alkaline, one of the hydrogen atoms (white hemispheres) starts to break off (disassociate). In the picture below, it is the hydrogen at the bottom left.
As the pH approaches neutrality (pH 7) more and more of the indicator molecules lose this hydrogen atom. At pH 7, half the litmus molecules will still have this hydrogen in place and the other half won’t. This mixture produces shades of purple in the mid-range. Once the solution goes to alkaline pH 8.3 or higher, all the hydrogen will have disassociated and the litmus indicator will now be blue.
Video: Using Litmus Paper for Simple Identification of Acids & Bases
Litmus Paper vs pH Strips
Since litmus paper gives indeterminate results around neutral pH 7 one has to consider alternative tests in this range. A pH meter is one option but this is relatively expensive and not recommended for science fair or homeschooling use.
pH test strips use different chemical indicators. Some have just one indicator that reacts in a narrow range such as pH 3-6. Other test strips use several indicators which can work with a wider range of pH levels. You can even choose between strictly base pH 7-14 strips or acid pH 0-7 strips. Or, use ones that cover a very wide range of acids & bases such as the pH 1 to 14, or just the ones on either side of neutral like the pH 4.5-10 test strips.

Try these combination packs of pH & litmus test strips. We have put together 3 different bundles that include a vial each of 100 blue and 100 red litmus papers and some different pH strips.
pH/Litmus Choices
You can do interesting experiments with these litmus paper and pH strip combination packs; see the list in the 3rd of pH/litmus paper.
There are also pH test strips for some foods and beverages such as:
- pH 3-6 acid test strips are good for sushi rice and salsa.
- At the basic end are pH 10-13 strips for maple syrup makers.






What if the test subject is solid?
Good question. Just wet the paper/strip, preferably with deonized or freshly distilled water & press against the object. If acid or base is present, it should cause a color change.
Many things change color with pH. Blueberries, purple cabbage, red onion skin, turmeric. My favorite is black bean soak water. Paper, wettened and dried with this behaves similar to litmus.
Yes and a good topic for science fair. But, if you’re in a hurry, these will do nicely.